  The intrinsic nature of the prevailing molecular structure includes either conjugated or unconjugated backbone of the molecule, molecular confirmation and similar substitution group apart from the backbone.Ĭonversely, molecule electrode contact is influenced by the anchoring groups, electrodes and contact configuration of the electronic that coupling amidst the metal and molecule. The answer to the question above of B: e Amines (e) are the least acidic acids of the group drawn above: amide anions (R2N-) are the least stable of the conjugate. Similarly, factors which facilitate the breaking of the X-H bond will also increase the acidity of HA. Ideally, the conductance of the single-molecule junctions is frequently influenced by the underlying intrinsic nature of the molecular structure and corresponding molecule electrode contact. equilibrium of this process and result in a smaller pKa (i.e. It entails two parts of coating the 11-Mercaptoundecanoic acid onto the nanotip and attaching the alcohol to the 11Mercaptoundecanoic acid.Ĭomprehending the charge transport of the single metal- molecule junctions is significant in the determination of the prospect molecular electronic devices (Hassner, Namboothiri & Hassner, 2012). The experiment aims at attaching carboxylic acid mainly 11-Mercaptoundecanoic acid employed to connect to conjugate the useful molecule to the nanotip. 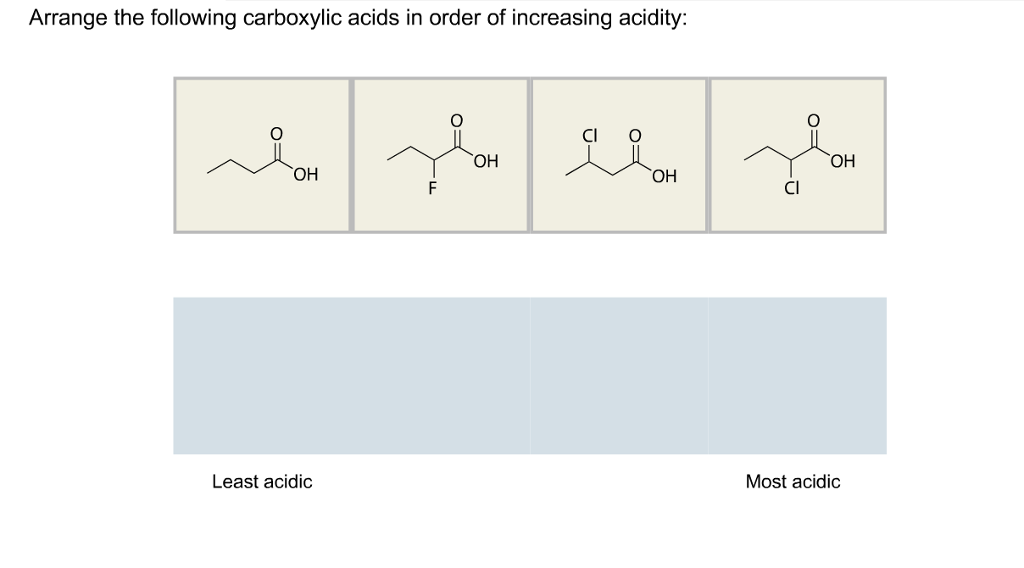 The product results in the structure subsequent to the experimentation are the regular confirmation via the Refractive Index, FTIR, and HNMR analysis. As you should know, acidity stems from the ability of a compound to release protons in the medium, so acids are proton donors. HClO Part B Arrange the following carboxylic acids in order. The ranking of these acids in order of increasing acidity is: CH3CH2COOHrank items as equivalent, overlap them. From 1 being the least acidic and 4 being the most acidic, arrange the following organic compounds in terms of increasing acidity. Start your trial now First week only 6.99 arrow. The alcohol mixes with the acid to undergo esterification, which entails a unimolecular two-step procedure of conversion via conjugation mechanism. Part A Arrange the following oxoacids in order of decreasing acid strength. Solution for Arrange the following types in order of increasing acidity: carboxylic acids, alcohols, phenols, water. The final Esters 03†and the final Esster04†both show multiple products eluting, and several show typical fragmentation for an alkyl chain containing the group. The conductance of the junction is relatively older as compared to the -(methylthio) benzoic acid junction as depicted in the experiment. Because of their enhanced acidity, carboxylic acids react with bases to form ionic salts, as shown in the following equations. The carboxylic acid is less steady since the CH3 group connected to the COO group that is destroyed by the mechanical steadiness of COO–linking.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
